
Solutions for better treatment of aortic aneurysms step closer
Geleen, March 21, 2024.
TripleMed BV is a medical startup focused on improving the treatment of aortic aneurysms. A consortium of existing and new investors has contributed more than two million euros to enable the clinical trials needed for CE marking and market launch. The aim is to achieve CE marking early in 2025. LIOF previously invested from the Limburg Business Development Fund (LBDF) and has now taken a stake in the company through the Participation Fund. Existing shareholder Brightlands Venture Partners (BVP) reinvested in TripleMed from its Chemelot Ventures fund.
Lenn Houbiers, investment manager at LIOF: “The TripleMed solution will eventually lead to more efficient management of aortic aneurysms, better quality of life and lower healthcare costs. In doing so, TripleMed makes an important contribution to the health transition, one of the transitions on which LIOF is strongly focused.”
Solutions under development
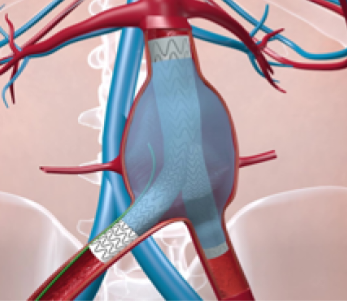
TripleMed is currently conducting clinical studies at a number of hospitals in the Netherlands and Belgium to validate AneuFix Endoleak Repair (fixing a leak after aneurysm surgery) and AneuFill Prophylactic Sac Filling (preventive insertion of a polymer to prevent leaks). The AneuFix/AneuFill concept is a 2-component polymer in a syringe. Upon insertion, the 2 components are mixed and then harden into an elastic permanent implant in the aneurysm.
Clinical trials started in 2020, 33 patients have been treated to date. The interim results of the clinical trial are very positive, in 89% of the patients the leakage remains stopped for a long time. By the end of 2024, the company hopes to have treated 57 patients and thus completed the clinical study so that CE marking can be obtained for AneuFix, followed in 2026 by CE marking for Aneufill.
“We are pleased with the new financial injection that will allow us to continue our research and achieve the certification required for the market launch of both products” said Tjeerd Homsma, CEO of TripleMed.”
Extent of aortic aneurysms
More than 150,000 patients worldwide are treated for aortic aneurysms each year. As many as 10-15% of all aortic aneurysms previously treated with stent-grafts experience leakage and further growth of the aneurysm. No effective treatment currently exists for this.
About TripleMed
TripleMed was founded in 2011 by three reputed vascular surgeons, Dr. Hans Brom, Dr. Alexander de Vries and Prof. Dr. Michael Jacobs. The company is based at the Brightlands Chemelot Campus in Geleen. TripleMed focuses on developing innovative and cost-effective solutions for the treatment of aortic aneurysms.
Despite the obvious benefits of endovascular treatment using endoprostheses, the procedure is associated with a relatively high number of complications and repeat operations in the years following the initial surgery, which has a major impact on patients’ quality of life and high costs. With its products, TripleMed expects to make a significant improvement to more effective and cost-efficient treatment of aortic aneurysms.
More information: www.triplemedical.com.
About Brightlands Venture Partners
Brightlands Venture Partners (BVP) is the fund manager of Chemelot Ventures and is a so-called ecosystem investor. BVP invests in companies benefiting from and contributing to the Brightlands campuses in the south of the Netherlands. Other funds under management are BVP Fund IV, Brightlands Agrifood Fund and Limburg Ventures. Chemelot Ventures has a portfolio of investments in startups and scaleups in sustainability and health. Together the BVP funds have made over 50 investments.
More information on https://brightlandsventurepartners.com/.

Image on the left: During the procedure, AneuFill polymer is inserted through a filling catheter immediately after the endoprosthesis is placed (green).
Image on the right: The entire space of the aneurysm around the endoprosthesis is filled with AneuFill polymer (blue).